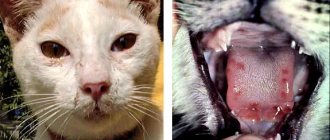
Many types of yeast fungi are opportunistic microflora that constantly live on the skin and mucous membranes of the animal’s body. They do not cause pathology and live in symbiosis with the host until a sharp decrease in immunity occurs. During this process, microorganisms begin to multiply intensively, which leads to the occurrence of diseases. A striking example is the development of Malassezia in cats.
Malassezia globosa fungus under a microscope
Malassezia dermatitis is caused by the yeast Malassezia pachydermatis. For the most part, they inhabit the external auditory canal, interdigital folds, anal glands in males, vagina in females, and the rectum. Only weakened animals suffer from this disease; with a timely diagnosis, treatment initiated and proper care, recovery occurs in a short period of time.
Causes of the disease
Malaceziosis is, as a rule, a secondary disease that appears against a background of weakened immunity. The reasons for this may be the following:
• infectious disease;
• food allergies;
• external and internal parasites;
• hormonal imbalance;
• diseases of the digestive or urinary systems;
• mechanical damage to the skin (bites, scratches, wounds).
The Malassezia fungus is considered opportunistic. It is found in small quantities on the skin of all cats and does not cause disease under normal conditions. When the protective barriers are weakened, caused by allergies, injuries, infections or other reasons, it is activated and begins to multiply intensively. As a result, the lipid-carbohydrate balance of the skin is disrupted and a disease called malasseziosis occurs.
Allergy to yeast of the genus Malassezia in patients with atopic dermatitis
Lipophilic yeasts of the genus Malassezia, which live on human skin, have unique properties that particularly distinguish them from the kingdom of fungi. The uniqueness lies in the fact that they represent a striking example of the ability of a microorganism, depending on environmental conditions and the host’s immunity, to exhibit the properties of a commensal or a pathogen. In addition, this is the only representative of the human microflora that requires fats for its vital functions. None of the other types of mushrooms have the quality of obligate lipophilicity.
History of the issue. Almost every person's skin, especially the upper part of the body, is colonized by fungi of the genus Malassezia. It would seem that this is why the study of the physiology of this constant companion of the human body should be comprehensive. Meanwhile, the evolution of studying the characteristics of cohabitation between host and fungus for more than a century was accompanied by many errors and inaccuracies. They were first described in 1846 by the microbiologist Eichstedt [1]. After this, for a long time the study of the physiological properties of these fungi was difficult due to the fact that it was not possible to cultivate them. These yeasts did not survive on media that were usually used for cultures of other fungi. It was only when Dr. Benham realized in 1939 that fats were necessary for the life of these fungi that it became possible to obtain a culture of Malassezia spp. [2]. The difficulty of identifying species was also due to the fact that fungi of the genus Malassezia are characterized by dimorphism, that is, the ability to remain in both the yeast and mycelial phases. Therefore, many researchers assumed that these phases are nothing more than different representatives of the yeast flora: the genus Pityrosporum is the yeast form, and Malassezia is the mycelial form. In addition, the variability in the shape of yeast was taken by microbiologists to be different species: the round shape of the cell is Pityrosporum orbiculare, and the oval shape is Pityrosporum ovale. Thus, the taxonomy and nomenclature of the genus Malassezia has been confusing and chaotic until recently [3, 4]. Due to the fact that the presence of two phases of yeast life was never proven, both genera were present in the classification. This situation became clear only in 1977, when three independent groups of mycologists published data that they were able to create conditions under which yeast cells produced hyphae in vitro. Using different variations in culture conditions, they were able to obtain hyphae that were different from the strains isolated from patients with lichen versicolor. Hyphae have been shown to be capable of producing both round and oval shapes of yeast cells, so it has been suggested that cell shape is a phase of the microorganism's life cycle.
Thus, progress in the knowledge of fungi of the genus Malassezia only by 1996 allowed the authors Guillot and Gueho to establish order in taxonomy [5]. They recorded 104 strains of Malassezia spp., identified by DNA signature determined by polymerase chain reaction methods. Based on these data, they identified and named 7 species of the genus Malassezia: M. furfur, M. sympodialis, M. obtusa, M. globosa, M. restricta, M. slooffiae and M. pachydermatis [6].
Biology, physiology, ecology of Malassezia spp. In connection with the problems of cultivation, biology, physiology, ecology of Malassezia spp. poorly researched. The main distinguishing feature of this yeast is its inability to ferment sugars. The main source of carbon for them is fats. Although the microorganism can be grown under aerobic conditions, it can also survive under anaerobic conditions. It is assumed that fatty acids are not a source of energy and do not participate in metabolism, but are an integral part of the cell. The lipids found inside the epidermal cells have the composition necessary to nourish yeast cells [7]. Until recently, the difficulty of obtaining cultural extracts did not allow studies of IgE-mediated reactivity in vivo and in vitro. And only in the last 20 years has it become possible to determine the role of the fungi Malassezia spp. in the pathogenesis of atopic dermatitis (AD).
Antigenic composition of Malassezia spp. The rich antigenic structure of lipophilic fungi is credited with the property of high immunogenic activity, which significantly exceeds that of other representatives of the yeast flora, such as, for example, Candida albicans. The ability of an antigen to cause the induction of antibodies in the human body has an extremely wide polymorphism. It depends both on the state of the host’s immune system, on the antigenic composition of fungi, and on the characteristics of the human environment (climate, composition of water, detergents, insolation, temperature, qualitative composition of the sebaceous secretion of the skin glands). Currently, most of the known antigens of the main species of the genus Malassezia have been described, their biochemical structure, the ability to induce IgE antibodies in the blood of patients with atopic dermatitis have been studied, and 13 main recombinant allergens have been obtained [8, 9].
A special type of immune response, different from that of non-allergic individuals, to Malassezia spp. antigens. is formed throughout life in patients with AD. One of the most significant allergens for sensitizing the body of a patient with AD is mannan of lipophilic fungi, which causes the induction of IgE antibodies specific to it and a positive prick test in half of the patients sensitized to Malassezia spp. Numerous studies have shown that in patients suffering from AD, but not rhinitis or asthma, IgE antibodies to fungi of the genus Malassezia are induced [10]. Several basic biochemical studies have shown high cross-reactivity between antigens of Malassezia spp. and other yeast-like fungi such as Candida albicans, Rhodotorula rubra, Cryptococcus albidus and Saccharomyces spp. It is due to the homology of antigenic determinants of high molecular weight mannoproteins isolated from these fungi. It is believed that primary sensitization is caused by the fungi Malassezia spp. [eleven].
The release of allergens on the skin is influenced primarily by the pH on the skin surface, which is higher in patients with AD than in healthy people. It turned out that M. sympodialis cells produce, express and release allergens in large quantities at the highest pH values. This is especially true for the 67-kDa major allergen, designated Mala s 12 [12].
Immune response to Malassezia spp . There are two conflicting points of view on the phenomenon of induction of IgE antibodies to yeast antigens, due to the proven fact that the levels of specific IgE to Malassezia correlate with the level of total immunoglobulin E, but do not depend on the severity of AD. A number of authors believe that the induction of lipophilic antibodies to mannan is only a manifestation of atopic status, since 85% of patients with AD have a significantly increased level of total IgE, and is not evidence of the role of these microorganisms in the pathogenesis of AD. Others, on the contrary, believe that this is the most important trigger factor for blood pressure.
It is assumed that autoreactivity to human proteins in patients with AD plays the role of one of the most significant pathogenetic factors in AD. One of these proteins, the enzyme manganese superoxide dismutase (SDM) - MnSOD, is induced under stressful situations by both human cells and fungal cells. It has been proven that the enzyme has the property of allergenic activity (autoallergen in AD). Molecular mimicry leading to cross-reactivity, such as sensitization, to MnSOD may be primarily due to colonization of the skin of patients with lipophilic fungi [13].
Thus, lipophilic yeasts of the genus Malassezia, which constantly live on human skin, begin to play the role of a strong antigenic stimulus in patients with AD. If the colonization of the skin surface by fungi occurs in the first years of a person’s life, then in the presence of an atopic predisposition, it is one of the first allergens that causes an immediate type of allergic reaction. Considering the cross-reactivity with other yeast-like fungi, which are constantly present on the mucous membranes, ingested every day with food, with identical enzymes produced by skin keratinocytes, lipophil producers become the main trigger and pathogenetic factors of AD throughout life.
Diagnostics. To diagnose AD complicated by skin colonization with Malassezia spp., mycological, immunochemical, and allergological methods are used.
Mycological research for the purpose of isolating and identifying lipophilic yeasts of the genus Malassezia is carried out by scraping and collecting from the surface of the skin in an area of 1 cm2 on a cotton swab. Next, subculture is carried out in triplicate on Notman-agar (LNA) selective medium: (10.0 g polypeptone, 5.0 g glucose, 0.1 g yeast extract, 8.0 g bovine bile, 1.0 mg glycerol, 0 .5 g glycerol stearate, 0.5 mg Tween 60, 10 ml milk and 12.0 g agar per liter). The inoculated dishes are incubated in a thermostat at 32 °C for two weeks. Species identification of representatives of the genus Malassezia is carried out according to morphological (colony morphology, cell size and shape), physiological (growth at 37 °C, 40 °C) and chemotaxonomic (catalase reaction, utilization of Tween 20, Tween 40, Tween 60, Tween 80) characteristics . The calculation of average CFU/cm2 values is carried out in accordance with the number of colonies grown on a plate from a sample taken from a 1 cm2 area of skin.
According to our data, all patients suffering from AD that we examined, including children, as well as approximately 70% of healthy patients without skin diseases, secrete yeast-like lipophilic fungi of the genus Malassezia. The number of yeasts of the genus Malassezia ranges from 10–106 CFU/cm2. The abundance of yeast isolated from the skin of the upper body is typically one to three orders of magnitude higher than that in samples obtained from healthy patients. On average, the number of Malassezia was 104–105 CFU/cm2 in patients and 102 CFU/cm2 in healthy people.
We were able to identify four species of the genus Malassezia: Malassezia sympodialis, Malassezia globosa, Malassezia furfur, Malassezia restricta. The dominant species in all studied samples was Malassezia sympodialis. We regularly isolated it from the skin of both sick and healthy people. The second place in occurrence is occupied by Malassezia globosa and Malassezia furfur.
Detection of IgE antibodies to Malassezia spp. in the blood serum of patients, it is recommended to carry out the immunochemiluminescence method on the UniCAP 100 device (Phadia, Sweden) using appropriate reagents. We detected IgE antibodies to Malassezia spp. in almost 40% of patients and in none of the healthy patients ( Fig.).
The average values of IgE-AT concentration in blood serum varied from 0.36 kU/l (class 1) to 68.8 kU/l (class 5). They were most often detected in patients older than 7 years; in three patients aged 5 years they were already present in significant concentrations (more than 0.36 kU/l). However, the concentration of IgE antibodies in the group of children was significantly lower (from 0.36 kU/l to 32.0 kU/l, average value 8.06 ± 7.9 kU/l) than in the group of adults (from 0.39 kU/l to 68.8 kU/l, average value 19.42 ± 16.7 kU/l) (p < 0.05).
Thus, although in our study IgE antibodies to Malassezia spp. were detected in only 40% of patients, clinical signs of sensitization to yeast-like fungi were noted in all patients included in the trial according to the relevant criteria. The presence and concentration of antibodies did not depend on the severity of AD. It has been previously shown that in atopic patients suffering from AD, but not rhinitis or asthma, IgE antibodies to Malassezia fungi are induced, so most researchers believe that this is the most important trigger factor for AD.
Thus, the presence of IgE antibodies to fungi of the genus Malassezia in the blood serum of patients with AD is not a sign that determines the severity of the disease, but is a marker of the chronic course of AD. It is very unlikely that during the “atopic march” the appearance of IgE antibodies to fungi of the genus Malassezia in the blood of children will accompany the transformation of AD into rhinitis or asthma with complete remission of dermatitis. The type and abundance of Malassezia yeast on the skin does not affect sensitization to these fungi, that is, there is no dose-dependent effect between the level of antibodies and the degree of fungal colonization on the skin. The data obtained show that the species and quantitative composition of lipophilic fungi living on the skin does not affect the pathogenetic mechanism of AD.
Treatment of AD with skin colonization by Malassezia spp.
Characteristic features of the clinical picture of AD in patients with skin lesions of the upper body are the connection of exacerbations with respiratory infections, the use of foods containing yeast-like fungi and easily digestible carbohydrate foods in the diet, white dermographism of the skin, low therapeutic effect of antihistamines and topical corticosteroids, severe course diseases. The majority of our patients (63%) associated the exacerbation of the disease with eating foods containing sucrose, starch, and flour after more than 24 hours. Nine adult patients noted that when drinking alcoholic beverages, mainly wine and champagne, they experienced severe flushing and itching of the facial skin for 2–5 hours. In all children, the disease worsened significantly after eating food containing yeast (bread, baked goods) and sugar for 24 hours. In this regard, the most significant in the treatment of patients sensitized to yeast is following a diet with a reduced content of easily digestible carbohydrates, foods that promote fermentation, and, most importantly, yeast-containing ingredients. These include, first of all, flour products containing baker's yeast Saccharomyces spp., beer, kvass, champagne, and wine. Yeast-like fungi, such as Rhodotorula spp., are found in almost all canned juices, canned vegetables, and seafood. Increased sugar content in the gastrointestinal tract promotes fermentation and the proliferation of yeast of the genus Candida.
Successful attempts at controlled clinical trials of systemic antifungal drugs in the treatment of patients with AD leave no doubt that the pathology of the immune response of atopic patients to fungi, commensals of human skin, is one of the most significant factors in the pathogenesis of AD. That is, reducing the concentration of antigenic yeast molecules in the body that can cause the induction of IgE antibodies leads to a very noticeable therapeutic effect. But the question of tactics for managing patients with AD remains open. Elimination of yeast from the body is not the final goal of treatment, since in the absence of resident skin flora the risk of infection by pathogenic microorganisms increases significantly. The true solution to the problem is to find that “golden balance” in which, even taking into account the induction of IgE antibodies to yeast, no inflammation of the skin is observed, that is, a symbiotic balance is achieved. It is advisable to prescribe antimycotics (Orungal, Diflucan) only in short courses, during exacerbations, in order to short-term reduce the allergenic exposure to the atopic organism. There are many studies in which the selection of doses and duration of courses of treatment with systemic antifungal drugs was carried out. It is suggested to administer fungicidal drugs in low doses over a long period of more than 6 months. So, for example, Orgungal is prescribed 200 mg per day in 1-2 doses for 2 weeks. If there is no positive effect after 2 weeks, treatment should be continued for another 2 weeks. However, side effects from long-term use of systemic antimycotics are a serious problem that cannot be ignored when prescribing such a prolonged course. That is why, despite successful attempts at treatment with fungicides, these drugs have not yet been included in the standard treatment of atopic dermatitis. And there is no consensus on this issue in the literature yet [14, 15].
The use of local ointment preparations with an antimycotic effect, such as terbinafine, clotrimazole, zinc pyrithione and others, gives a less pronounced but still positive therapeutic effect. Their inclusion in the treatment of blood pressure is undoubtedly an important component, since these drugs do not have the systemic toxicity and withdrawal syndrome characteristic of topical steroids. In addition, we should not forget that daily body care should include cosmetics for the care of the scalp and shower gels with mild fungicidal activity (Nizoral shampoos, Friederm tar, Skin-cap), which patients should use regularly to reduce the number of lipophilic yeasts on the skin.
Conclusion
Recently, questions about the polymorphism of genetic and sensitizing factors in the immunopathogenesis of AD are increasingly discussed in the periodical literature. The absence of IgE-mediated reactivity in 20% of patients with AD with completely similar clinical symptoms suggests a combination of complex reactions, including an inadequate immune response to skin commensals such as Malassezia spp. and Staphylococcus aureus. The unique properties of lipophilic Malassezia fungi allow them to be present on the skin of the human body throughout life almost unhindered. Their protective role as an antibacterial and antifungicidal agent, as an ultraviolet filter and tumor depressant can hardly be overestimated. The striking property of a suppressive and stimulating effect on the immune system makes it possible to experience various polymorphic unfavorable environmental conditions and the condition of the person himself. Skin lesions of the face, neck and shoulders are a characteristic sign of hypersensitivity to Malassezia spp., which is observed in approximately 30-40% of all patients with AD. Thus, sensitization to fungi of the genus Malassezia is one of the most significant trigger factors in the pathogenesis of AD.
Literature
- Eichstedt E. Pilzbildung in der Pityriasis versicolor. Frorip Neue Notizen aus dem Gebeite der Naturkunde Heilkinde. 1846, no. 39, p. 270.
- Benham RW The cultural characteristics of Pityrosporum ovale–a lipophilic fungus // J. Investig. Dermatol. 1939, no. 2, p. 187–203.
- Panja G. The Malassezia of the skin, their cultivation, morphology and species. Trans. 7th Congr. Far East. Assoc. Trop. Med. 1927, no. 2, p. 442–456.
- Slooff WC Pityrosporum sabouraud. In J. Lodder (ed.): “The yeasts—a taxonomic study,” 2nd ed. North Holland Publishing Co., Amsterdam, The Netherlands. 1970, p. 1167–1186.
- Guillot J., Gueho E. The diversity of Malassezia yeasts confirmed by rRNA sequence and nuclear DNA comparisons. Antonie Leeuwenhoek. 1995, no. 67, p. 297–314.
- Ashbee HR Update on the genus Malassezia // Med Mycol. 2007. V. 45, No. 4, p. 287–303.
- Wilde PF, Stewart PS A study of the fatty acid metabolism of the yeast Pityrosporum ovale // Biochem. J. 1968, no. 108, p. 225–231.
- Koyama T., Kanbe T., Ishiguro A. et al. Antigenic components of Malassezia species for immunoglobulin E antibodies in sera of patients with atopic dermatitis // J Dermatol Sci. 2001, v. 26, no. 3, p. 201–208.
- Johansson S., Karlstrom K. IgE binding components in Pityrosporum orbiculare identified by an immunoblotting technique // Acta Dermato-Venereol. 1991, v. 71, p. 11–16.
- Zargari A., Midgley G., Back O. et al. IgE-reactivity to seven Malassezia species // Allergy. 2003, v. 58, no. 4, p. 306–311.
- Huang X., Johansson S.G.O., Zargari A. et al. Allergen cross reactivity between Pityrosporum orbiculare and Candida albicans // Allergy, 1995, v. 50, p. 648–656.
- Selander C., Zargari A., Mollby R. et al. Higher pH level, corresponding to that on the skin of patients with atopic eczema, stimulates the release of Malassezia sympodialis allergens // Allergy. 2006, v. 61, no. 8, p. 1002–1008.
- Schmid-Grendelmeier P., Flickiger S., Disch R. et al. IgE-mediated and T cell-mediated autoimmunity against manganese superoxide dismutase in atopic dermatitis // J. Allergy Clin Immunol. 2005, v. 115, 5, p. 1068–1075.
- Back O., Scheynius A., Johansson SGO Ketoconazole in atopic dermatitis: therapeutic response is correlated with decrease in serum IgE. Arch. Dermatol. Res. 1995, v. 287, p. 448–451.
- Clemmensen OJ, Hjorth N. Treatment of dermatitis of the head and neck with ketoconazole in patients with type I sensitivity to Pityrosporum orbiculare. Semin. Dermatol. 1983, no. 2, p. 26–29.
M. A. Mokronosova, Doctor of Medical Sciences, Professor of the I. I. Mechnikov Research Institute of Internal Medicine, Russian Academy of Medical Sciences, Moscow
Diagnostics
The diagnosis of malasseziasis is made based on the following examination results of the animal:
• medical history data – the patient’s living conditions and illnesses he has suffered;
• identifying characteristic symptoms of the lesion;
• detection of a large number of Malassezia cells in skin scrapings under a microscope and colonies of the fungus during culture.
Most often, malacesia occurs in weakened cats or in kittens during the transition from breastfeeding to solid food .
During the examination, if characteristic symptoms of the disease are detected, scrapings are taken from the affected areas of the cat’s skin, which are examined under a microscope. At the same time, inoculations are made on the nutrient media.
Under a microscope, Malassezia looks like a groundnut. The cells are arranged in groups of several, attached to a nutrient substrate - the epidermis or hairs of wool. The diagnosis of malassezia is made if more than 10 microorganisms are detected in 15 points of the test sample at 1000x magnification. When sown on Sabouraud agar, Malassezia forms convex, well-defined, spherical colonies of yellow or yellowish-brown color.
Since malacesia is a secondary disease, in parallel with cytological studies, tests are carried out for the presence of allergies, and blood is taken for biochemistry. If there are no results, a comprehensive examination of the animal is carried out to identify the main cause of the disease.
Therapy
Treatment of Malassezia in cats can be carried out in several ways. But let us warn you once again that before starting therapy it is very important not only to identify the pathogen, but also to make sure that it is the one that caused the disease. If treatment over a long period of time does not produce a positive effect at all, a full examination of the animal should be carried out in order to identify the true root cause of the pathology.
To create an unsuitable environment for the fungus to live, it is necessary to remove excess fat from the cat's skin. For this, both specially developed shampoos and more conventional products can be used. A 1% solution of chlorhexidine has proven itself well. In severe cases, the concentration may be increased. Products containing benzoyl peroxide and sulfur are also used.
Classification of fungal diseases
Diseases caused by pathogenic fungi are divided into 2 groups depending on the location of the lesion:
- superficial mycoses, the causative agents of which parasitize the skin and mucous membranes of animals;
- systemic mycoses that cause pathological changes in internal organs.
Diagnosis of the second group of diseases is difficult due to the lack of clinical symptoms characteristic of common fungal infections. Therefore, even signs of a cold or intestinal disorder should be a reason to contact a veterinary clinic, where an experienced doctor will be able to recognize the exact cause of the disease.
The most frequently reported fungal diseases include:
- ringworm caused by parasites of the genus Trichophyton and Microsporon;
- Malassezia, caused by the ear fungus Malassezia;
- aspergillosis, caused by saprophytes of the genus Aspergillus;
- candidiasis provoked by yeast fungi of the genus Candida;
- cryptococcosis caused by the saprophytic fungi Cryptococcus.
Once pathogenic microbes have reached the skin surface, they immediately begin to harm the health of the cat (by releasing toxic toxins and enzymes).
Over time, this can lead to the death of the epidermis from the upper skin and the development of inflammatory processes.
After all, without proper care, the fungus will penetrate deeper into the pet’s body, damaging the fur structure and hair follicles.
What signs can be used to determine the presence of harmful parasites? First of all this:
- Loss of damaged pieces of fur;
- The presence of empty bald spots on the cat with clear boundaries in the head, ear area, and also on the paws;
- Restless behavior (the animal runs from corner to corner, itches and often rubs against objects);
- Scabies;
- Bad smell;
- The presence of a strange, pinpoint plaque in the ears;
- Formation of tubercles on the skin surface;
- Frequent sneezing;
- Bloody runny nose.
If the situation is neglected, the initial symptoms can develop into a severe form of the disease, which will lead to anemia, pneumonia, and complications in the gastrointestinal tract.
If the cat is still young or weakened (for example, due to lack of food), this can be fatal.
Malassezia in animals: what are the advantages of veterinary
Malassezia should be treated according to the recommendations of a specialist - a veterinarian. Before turning to any veterinary center for help, it is recommended to soberly evaluate its advantages and decide for yourself whether it is good enough for you. Veterinary, has the following advantages:
- Modern equipment, both in the veterinarian’s office and in the laboratory;
- Qualified specialists who regularly update their knowledge with new knowledge;
- Positive attitude towards animals;
- Possibility of calling a doctor at home;
- Possibility of testing at home;
All these advantages set us apart from other veterinary centers and allow us to fully experience all the delights of a modern approach to the provision of veterinary services.
Allergic skin diseases
Many breeders associate the word “allergy” with a runny nose, watery eyes and other minor troubles. This is partly true, but allergic reactions in practice are much more dangerous. In particular, they are considered the main cause of incurable and deadly diseases, but even in “moderate” cases of pathology, these pathologies can cause a lot of trouble.
Allergic dermatitis
Dermatitis is a pathology accompanied by inflammation of the skin. Accordingly, in this case, the inflammation is caused by a serious allergic reaction.
Firstly, the latter leads to a catastrophic weakening of the skin’s protective mechanisms, which is why it can no longer resist the action of pathogenic and conditionally pathogenic microflora.
Secondly, a much worse option is possible: due to increased vascular permeability, lymphocytes begin to migrate en masse into the thickness of the epithelium. This effect is called lymphocytic or neutrophil infiltration and, by the way, it often leads to the development of autoimmune inflammation. Stress factors accelerate and intensify the pathological process.
Allergic eczema
In many ways, this disease is similar to the previous one, but still it differs in a number of specific aspects, which is why these pathologies should not be considered comprehensively.
Milliary dermatitis
One of the varieties of the previous pathology. However, this disease has specific features that make it possible to distinguish it into a separate subsection.
Recommendations
Animals suffering from malasseziasis are advised to periodically bathe with a special shampoo. You should monitor the folds of the skin, the external auditory canal, the interdigital areas, and if brown plaque appears, begin local treatments, since local treatment carried out on time can minimize the intake of tablets and medications for the cat.
You should not keep the animal constantly in a humid environment; you need to monitor its diet (if a food allergy is diagnosed). To protect your cat from fungus, you need to regularly treat the animal against fleas and other ectoparasites.
Sources
- https://NoParasites.ru/parazity-u-zhivotnyx/malasseziya-u-koshek.html
- https://MyFavoritePet.ru/zdorove/malasseziya-u-koshek-v-ushah.html
- https://blotos.ru/malassezia-u-kosek-v-usah-simptomy-lecenie-foto
- https://KotPoloskin.ru/zdorove/malasseziya-u-koshek-v-ushah-lechenie.html
- https://InfoKotiki.ru/zabolevania/kozhi/malasseziya-u-koshek.html
- https://probakterii.ru/fungus/malasseziya-u-koshek.html
- https://KotuDom.ru/zdorove/malassezionnyj-dermatit-u-koshek-2.html
- https://litbro.ru/veterinariya/vaktsinatsiya-koshek-ot-lishaya
- https://arcticat.ru/bolezni/malassezionnyj-dermatit-u-koshek.html
- https://vashipitomcy.ru/publ/zdorove/bolezni/malassezija_gribok_u_koshek_osnovnye_projavlenija_i_metody_lechenija/15-1-0-1185
- https://zoostatus.ru/lechenie/bolezni/malassezii-gribok-u-koshek/
[collapse]
How contagious is the fungus in Malassezia?
When doctors use the term “malassezia,” they mean a fungal organism of the yeast species. It is slightly convex in shape and has a cylindrical structure, smooth, soft, loose, and small (if translated into microns, it is two and a half microns). Looking at it, you may think that you are seeing an ordinary nut. Malassezia crops are characterized by an unusual smell with a “fruity” tint.
When you see someone with Malassezia dermatitis, you don’t have to run away in fear or hide your pet away. The idea that Malassezia fungus is contagious is completely false. Since it is one of the components of the microflora, which tends to be present on the epithelial tissue of the animal throughout its life.
In everyday life, the fungus causes absolutely no harm. Excessively fast and large reproduction begins after any incidents that create an excellent environment for this. Most often this is provoked by various changes in the microclimate of the skin (humidity, warmth, advanced inflammation).
Nutritional Features
When symptoms of atopy appear, the animal needs a special diet. You need to follow it for at least 2 months, after which you can carefully switch to the usual diet (if the cat eats natural food). Veterinarians advise switching the animal to rice and rabbit if its protein does not cause a negative reaction. Additionally, the pet should receive omega-3 polyunsaturated fatty acids and biotin.
On such a diet without treatment, symptoms will subside after 2-3 months. If it is supported by drug therapy, the cat gets better much faster. For animals that prefer ready-made food, a hypoallergenic holistic food is selected. With the consent of the doctor, you should give your cat fish oil, cleansing tea, and vitamins (for example, Vittri).
Is it possible to develop immunity to fungus?
Cats do not develop resistance to fungal forms of lichen after an illness. General immunity is produced exclusively by blood cells when a foreign agent penetrates the bloodstream.
Lichen fungi do not penetrate blood vessels and live on the surface of the skin and in the animal’s fur. Accordingly, a cat that has once had shingles can become infected again.
There is a theory about the presence of local (skin) immunity in the body of living beings. That is, skin leukocytes recognize antigens (in this case, fungi) and produce antibodies to them. Moreover, lichen fungi come into contact with the surface of the skin. It is on this fact that the assumption about the effectiveness of vaccinations against lichen is based.
Is vaccinating cats against lichen effective?
Today, only Russian vaccines for the prevention of lichen in animals are available in veterinary pharmacies. Abroad, such remedies have disappeared and are not used due to the low prevalence of the disease.
Since in Russia lichen is very often registered in agriculture, fur farming and among domestic pets, there is a demand for preventive biological products. However, to date, no pharmaceutical company has published reliable data or studies that support the effectiveness of vaccinating animals against zoster.
Science has not proven that antifungal vaccines can protect against possible infection. Even if you repeatedly vaccinate kittens or rabbits against lichen, upon contact with the pathogen, the animals still get sick and go through all stages of clinical development on a par with those who were not vaccinated.
The effectiveness of the vaccine was studied on a small group of experimental animals, the result was positive, so a vaccine against lichen exists.
In fact, biological products (such as vaccines) require larger studies and many animals with lichen (and pets are selected based on similar physiological parameters and the same stage of development of the disease), which is extremely difficult and financially expensive. Therefore, many experienced veterinary specialists are convinced that vaccinations against lichen are not a preventive measure. However, vaccine manufacturers have their own point of view on this matter.
Ear hematoma in cats
With a hematoma, blood flows from the blood vessels of the auricle into the tissue under significant pressure, pushing these tissues apart and forming a cavity. The size of the hematoma depends on the strength of blood pressure in the damaged vessel, as well as on the degree of compliance of the tissues located near it.
A hematoma occurs quickly and its volume increases until the pressure from the stretched tissue becomes equal to the pressure in the damaged blood vessel. After this, the spilled blood clots, and a blood clot forms in the blood vessel.
Most often, hematomas in cats occur on the inner surface of the ear and much less often on the outside. The damaged ear increases in size, hangs down, and the swelling is painful and hot to the touch. If the hematoma is not treated, the pain only increases, and the hematoma itself can become infected with secondary microflora, which can ultimately lead to necrosis of the ear cartilage.
During a clinical examination of such a cat, a veterinarian notes the following symptoms:
- We observe anxiety and nervousness in the cat.
- The cat almost constantly shakes its head from side to side.
- He constantly scratches his damaged ear with his paws.
- When trying to stroke the cat's head, it becomes aggressive.
Treatment. Treatment of auricular hematoma is not very difficult. If no more than 48 hours have passed since the formation of the ear hematoma, then the cat owner fixes the ears with a bandage on the back of the head and applies cold. In the future, in order to resolve the hematoma, it is necessary to use heat and apply locally irritating ointments.
If the hematoma cannot be cured at home, the owner needs to contact the nearest veterinary clinic. In a veterinary clinic, the veterinarian will open the resulting hematoma, remove blood clots from it, wash the resulting cavity with a solution of novocaine with antibiotics and give recommendations to the owner so that the hematoma resolves safely.
Clinical picture
What are the symptoms? Some animals do not show any symptoms throughout the course of the disease, with the exception of constant “over-grooming” - they constantly lick themselves, causing their fur to literally stick together and become shiny with saliva. But this still rarely happens: more often the infection makes itself felt through patches of inflamed, reddened and flaky skin that appears in different parts of the body of a sick animal. Generalized forms of fungal dermatitis are relatively rare, and this can only be seen in the case of weak, emaciated and seriously ill animals, whose immune system cannot resist the action of pathogenic fungi.
Typically, classic Malassezia dermatitis develops in animals between one and four years of age. Let us remind you once again about the long-suffering sphinxes - in their case, it is very easy to see the onset of the disease. A thin brownish sebaceous film appears on the animal's skin. There is especially a lot of it in the interdigital spaces. And from this, cats, by the way, begin to constantly chew and lick their paws. In addition, the accumulation of sebaceous exudate occurs especially actively in the groin area. The consequences are the same - a sick animal constantly licks and chews these places, trying to get rid of the burning and itching.
It is important not to miss this moment. If at this time you start bathing your pet regularly, once every two to three days, using a 0.2% fluconazole solution, the disease can be defeated in just a couple of weeks. Unfortunately, in representatives of long-haired breeds, the external manifestations of the disease are not so obvious, and therefore the disease gradually progresses.
Ringworm vaccines for cats
Today, Russian veterinary medicine has four vaccines in the form of injections, which are prescribed for lichen in cats, dogs and fur-bearing animals.
Vaccerm Vaccine for cats
The biological product is a dry or liquid mass with inactivated spores of dermatophytes (fungi of the genus Microsporum canis, Microsporum gypseum and Trichophyton mentagrophytes).
The drug Vakderm was created for the immunization of healthy cats, dogs, rabbits and fur-bearing animals, as well as for identifying infected animals. If the cat has previously been in contact with a shingles animal, but clinical signs have not yet appeared, then after vaccination the process will accelerate. But the vaccination will not harm a completely healthy cat and no signs of disease will appear.
According to the manufacturer of the Vakderm vaccine:
- the vaccination has a therapeutic effect, if it is given three times (1 time to detect the disease, 2 and 3 times to speed up healing when lichen has been detected), then after 15-25 days the animal will recover;
- After immunization, animals become immune to lichen for 1 year.
Recommended scheme for administering injections against lichen for cats:
- The first vaccination of kittens is permissible from 30 days of age, adult cats - at any age;
- repeat injection - after 14 days and wait 25 days until stable immunity is developed.
Injections are given intramuscularly at any time of the year.
You cannot vaccinate cats with Vakderm in the second half of pregnancy.
Possible reactions after vaccination: drowsiness of the cat for 2-3 days and painful swelling at the injection site (goes away after a few days).
Important! If a dry vaccine is purchased, the drug should be used within half an hour after dilution.
Vakderm F
This vaccine is exclusively for cats because it consists of inactivated feline ringworm spores. The drug is completely similar to the Vakderm vaccine in terms of the principle of action, regimens of use, and contraindications. It differs only in that it has a mono composition, while Vakderm consists of a mixture of lichen spores from different species of animals.
Vaccine Polivak-TM
This is a shingles vaccine for cats that includes 8 varieties of mushrooms. Specially adapted for domestic cats. The vaccine is produced by (Narvak).
Manufacturers recommend starting vaccination of healthy kittens at the age of 1-3 months, adult cats - at any age, twice, with an interval of 14 days. After 30 days from the last injection, cats develop immunity to microsporia and other forms of lichen, which lasts for 12 months.
If the animal lives in an unfavorable environment, then a year later a revaccination is carried out, which must be repeated annually.
When treating lichen, sick cats are vaccinated three times with an interval of 2 weeks. After 1 month the animal recovers.
According to the stated principle of action, Polivac is no different from Vakderm, only the manufacturers are different.
Microderm
The vaccine is produced by Microderm. The drug is more expensive in cost than previous drugs.
Microderm is intended for the treatment and prevention of dermatophytoses (from microsporia and trichophytosis) in cats, dogs, rabbits and nutria.
The method of administration of this vaccine is similar to analogues: a healthy kitten is vaccinated twice and repeated annually. Sick cats are injected with Microderm once into the muscle. After 2 weeks, a pronounced therapeutic effect is observed, which is manifested by the rejection of epithelial scales and the growth of new hairs.
Prophylactic use of the vaccine does not cause the development of the disease in healthy animals. Immunity develops approximately 3-4 weeks after the last vaccination.
The main provoking factors of Malassezia
In order to protect your dog from the disease and, if necessary, take immediate action, you need to know what factors may favor the active growth of fungus on the skin and diagnose possible concomitant diseases.
It is reliably known that the disease is not contagious. It is not transmitted from dog to dog. The disease has internal causes. Malassezia canine fungus is also not contagious to humans and other animals.
Many years of experience in breeding dogs allowed experts to draw a conclusion about the main reasons for the active proliferation (spread) of fungus on the skin of animals, including:
- weakened immune system;
- infectious diseases;
- chronic diseases;
- allergic reactions of various etiologies;
- any skin diseases that violate the integrity of the skin;
- taking hormonal drugs and antibiotic therapy;
- too frequent use of shampoos and other detergents;
- poor care for thick, long hair.
Weakened immunity
The main cause of the disease is weakened immunity. Low resistance to skin infections may be hereditary or may be a chronic disease. A hereditary disease is diagnosed very early, when skin diseases begin to attack the puppy at a very tender age.
Chronic diseases
Chronic diseases are no less dangerous, as they cause a persistent decrease in immunity. Constantly progressing, they create favorable conditions for the growth of colonies of the fungus - the invader. The dog's skin becomes a pathogenic environment for them, which they try with all their might to suppress.
Infectious diseases
Skin infectious diseases have the same result - immunodeficiency. In addition, many of them are characterized by severe, unrelenting itching. Skin scratched until it bleeds is a gateway for pathogens. And pathological organisms for the fungus are a sign of rapid reproduction in order to fight “for territory.”
Allergic reactions of various etiologies
An allergic reaction on the skin creates the same conditions (scratching, violation of the integrity of the skin, decreased immunity) that encourage microorganisms to actively multiply.
Treatment with hormonal drugs and antibiotics
Hormone replacement therapy in most cases has side effects. In particular, this has an adverse effect on the organs of the endocrine system. Their functions may be impaired, causing the production of natural hormones to suffer. All this does not have the best effect on the condition of the skin. And this is a direct path to activating fungal growth.
Taking antibacterial agents also has its downsides. By actively suppressing the growth of pathogenic bacteria, they at the same time deal a significant blow to the body’s protective functions. Therefore, each intake of antibiotics must be accompanied by a parallel intake of antifungal and immunostimulating agents.
Frequent use of detergents
One of the main factors of protection against fungal infection is maintaining the integrity of the skin. Frequently used shampoos and detergents can artificially disrupt this integrity. They wash away the protective layer of the epidermis, and the skin becomes dry and susceptible to various infections.
But at the same time, careful ear care is necessary. There is a favorable environment for the development of Malassezia - a warm and humid environment. The ear canals should be promptly cleaned of wax secretions, wiping them dry each time.
Poor grooming
Long, poorly combed hair creates an obstacle to good air exchange and ventilation of the skin. Humidity and high temperature of the skin make it possible for fungal infections to quickly multiply on it.